HEV Hepatitis E Virus IgM Rapid Test
【INTENDED USE】
The Hepatitis E Virus Antibody IgM Test is a rapid chromatographic immunoassay for the qualitative detection of IgM antibodies to HEV in Whole Blood/Serum/Plasma to aid in the diagnosis of HEV infection. The test is based on immunochromatography and can give a result within 15 minutes.
【TEST PRINCIPLE】
Hepatitis E is an acute, usually self-limiting disease of the liver caused by hepatitis E virus (HEV). HEV is transmitted from person to person, primarily by the faecal-oral route. The incidence of hepatitis E is closely related to socioeconomic development, and seroepidemiological studies show that prevalence of anti-HEV antibodies in the general population varies from 15% to close to 100% in different parts of the world.
【Reagents And Materials Supplied】
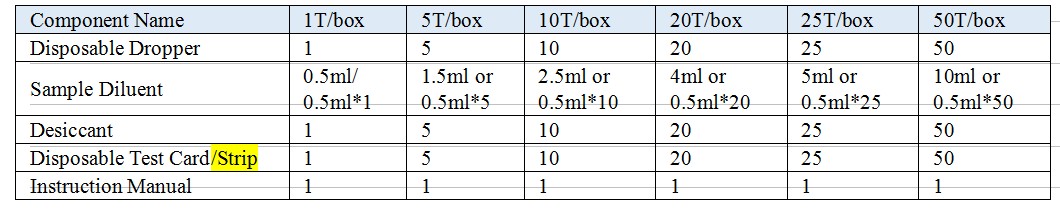
Model: Test Card, Test Strip
【SHELF LIFE AND STORAGE】
1. The original packaging should be stored in a dry place at 4-30°C and protected from light.
2. The shelf life of the test kit is 3 years from date of manufacture. Refer to the product labels for stated expiration date.
3. The original packaging can be transported at 2-37°C for 5-7days.
4. After opening the inner package, the test card will become invalid due to moisture absorption, please use it within 1 hour.
【Test procedure】
1.Bring the pouch to room temperature before opening it. Remove the test device from the sealed pouch and use it as soon as possible.
2.Place the test device on a clean and level surface.
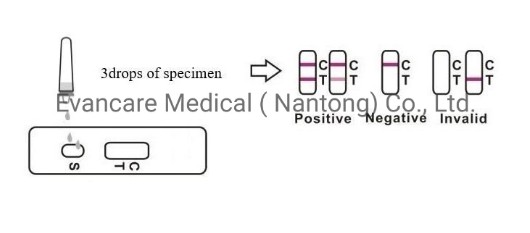
3.Hold the dropper vertically and transfer 1 drop of specimen (approximately 10μl) to the specimen well(S) of the test device, then add 2 drops of buffer (approximately 70μl) and start the timer. See illustration below.
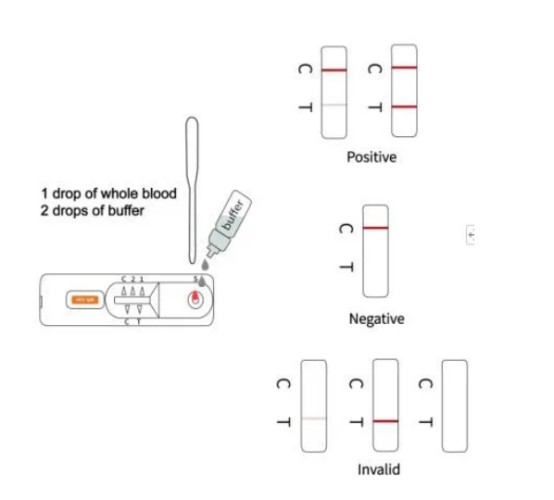
【INTERPRETATION OF ASSAY RESULT】
Positive: *Two lines appear. One colored line should be in the control region (C), and another apparent colored line adjacent should be in the test region (T).
Negative: One colored line appears in the control region (C). No line appears in the test region (T).
Invalid: Control line fails to appear. Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for control line failure. Review the procedure and repeat the test using a new test cassette.
If the problem persists, discontinue using the lot immediately and contact your local distributor.